Building Bruch’s Membrane to Better Understand Macular Degeneration
NewsThe Context: Age-related macular degeneration (AMD) results from the death of retinal pigmented epithelial cells (RPEs) in the eye. Bruch’s membrane is a structure that sits below RPEs and helps them receive blood — it also can become fragmented and less effective with age, which may factor into AMD. Creating a comprehensive stem cell model of AMD could benefit from effective models of Bruch’s membrane with which to study cell interactions.
The Study: A team of researchers including NYSCF scientists generated RPE cells from stem cells of patients with AMD as well as healthy controls and seeded these cells onto a substance designed to mimic an aged Bruch’s membrane, finding that the patient-derived cells recapitulated characteristics of AMD in this environment. The researchers also compared the patient-derived cells to healthy controls, finding that AMD-affected cells showed reduced metabolism. The study appears in Stem Cells Translational Medicine.
The Importance: This study demonstrates that a model of an aged Bruch’s membrane can be used in the laboratory to successfully recreate aspects of AMD, opening the door for more comprehensive studies of the disease. Identifying reduced metabolism in patient-derived cells also points to new avenues for study and treatment.
Age-related macular degeneration (AMD) affects 11 million Americans, causing loss of central vision which is currently incurable. The disease results primarily from the death of eye cells called retinal pigmented epithelial (RPE) cells, but many factors play a role in its onset and progression — including the aging and dysfunction of a structure called Bruch’s membrane.

A study in Stem Cells Translational Medicine by a team of researchers led by Mark Fields, MPH, PhD, of Yale University in collaboration with NYSCF scientists integrates a stem cell model of AMD with a model of Bruch’s membrane to better recapitulate features of AMD.
What is Bruch’s membrane?
Bruch’s membrane is a layer of tissue positioned below the eye’s RPE cells. It acts as a filter that regulates how much blood is delivered to the RPE layer. As we get older, Bruch’s membrane can become fragmented and calcified, and changes in its structure can precede AMD.
What did the researchers find?
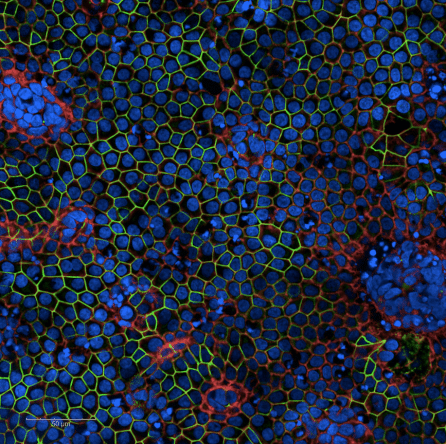
The scientists generated RPE cells from the stem cells of patients with AMD as well as healthy controls. They then seeded these cells in an extracellular matrix modified to resemble an aged Bruch’s membrane. The team found that AMD-affected RPE cells did not attach and survive to the model membrane as well as healthy controls, similarly to how cells in AMD behave. The researchers also discovered that patient-derived RPE cells showed reduced metabolism — a possible explanation for why they were less capable of attaching to the membrane model.
Why does it matter?
This study demonstrates that a model of aged Bruch’s membrane can be used to study AMD in the laboratory – opening the door for more comprehensive understanding of the disease. Additionally, uncovering differences in the cellular functions (such as metabolism) of healthy vs. AMD-affected RPE cells will point researchers toward new avenues for study and treatment of the disease.
Journal Article:
Stem cell‐derived retinal pigment epithelium from patients with age‐related macular degeneration exhibit reduced metabolism and matrix interactions
Jie Gong, Hui Cai, NYSCF Global Stem Cell Array Team, Scott Noggle, Daniel Paull, Lawrence J. Rizzolo, Lucian V. Del Priore, Mark A. Fields. Stem Cells Translational Medicine. 2019. DOI: https://doi.org/10.1002/sctm.19-0321

