All Your Questions About the Science of COVID-19, Answered – Plus How Stem Cells Can Help
News VideoFeatured image credit: National Institute of Allergy and Infectious Diseases
What makes the COVID-19 virus so hard to contain? Why do some people have severe reactions? What new treatments and diagnostics are on the horizon?
There are a lot of questions surrounding the COVID-19 pandemic, which has impacted millions worldwide. Last week, NYSCF Senior Vice President of Research Scott Noggle, PhD, presented a live research update – part of our new series of virtual events – to discuss the biology of COVID-19, how NYSCF is using stem cell models to study the infection, and the future of diagnostics and treatments.
What do stem cells have to do with COVID-19?
Since COVID-19 is a new disease — first described in November 2019 — there is still a lot to learn about it. A laboratory model for studying the virus and how it affects our cells is essential. Traditional laboratory disease models have critical limitations for studying COVID-19, including mice (who don’t have the protein ‘doorway’ that the virus uses to enter the cell), ferrets (whose immune response resembles that of young, healthy people), or cell lines (which can be infected, but don’t capture the immune response). With stem cells, scientists can create the actual cells that are susceptible to infection the way it occurs in patients, allowing them to examine how cells get sick and screen large numbers of drugs on infected cells.
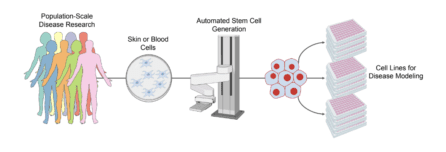
“Current models do not allow us to understand the biology of the infection or how it is impacted by genetics well enough,” explained Dr. Noggle. “Stem cells capture patient genetics, and if there’s a genetic component to a disease – and perhaps there is to COVID-19 – we can try to tease that apart. We study the disease process over and over in the laboratory, trying to figure out what the first thing that goes wrong is, and more importantly, how can we develop therapeutics to block that process?”
How do you catch the coronavirus?
SARS-CoV-2, the virus that causes COVID-19, is largely transmitted through respiratory droplets.
“When you cough, sneeze, talk, sing, or basically vocalize loudly, respiratory droplets come out of your mouth,” explained Dr. Noggle. “Studies show that in people infected with SARS-CoV-2, each one of these droplets can carry thousands to millions of viral particles.”
This is why it’s important to wear a mask when you go out in public. Masks serve as a protective measure to keep you from infecting others.
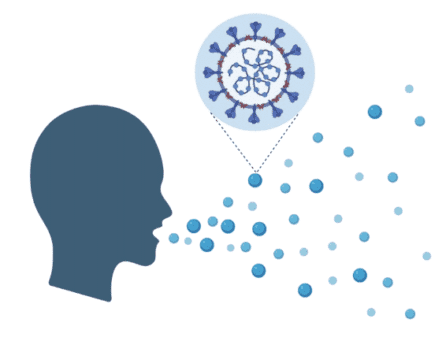
“Droplets get smaller as they travel. The cloth masks do a pretty good job of blocking large droplets, but they don’t block the small droplets as well. So, while they’re not perfect, they’re mainly meant to contain the virus to the person that’s wearing a mask and reduce spread.”
Once droplets are emitted, they fall or evaporate. But if they reach another person – say, for example, someone nearby inhales droplets, or someone touches a surface with particles on it and then touches their nose or mouth – then the virus can infect that person’s body. This is why recommendations to prevent transmission include social distancing and frequent hand washing as well as masks.
What happens once the virus gets into your body?
“Viruses are unique biological entities,” said Dr. Noggle. “They’re little machines whose sole purpose is to make more copies of themselves.”
But they can’t do this on their own: they need help from the cells in our bodies. Each virus has spike proteins on its exterior that it uses to latch onto the receptors of our cells. The virus then gets engulfed by the cell, where it then hijacks the cell’s machinery to make more of itself.
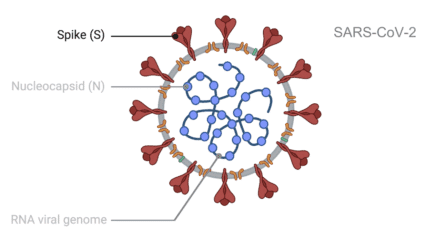
“It’s thought that once the virus finds a receptor on your cell, it takes about 10 minutes for that virus to make its way inside the cell and release its contents,” noted Dr. Noggle. “And it takes about another 10 hours for the hijacked cells to make thousands to millions of copies of the virus. And then, in most cases, the virus kills the cell as it bursts through, releasing all the replicates into the surrounding space outside the cell.”
Stem cell models are helping scientists to better study how exactly viruses infiltrate and affect different types of cells (e.g., lung cells, colon cells) that are susceptible to SARS-CoV-2 infection.
What makes the virus so hard to control?
A major challenge in diagnosis and containment of COVID-19 is that many infected people do not show symptoms right away — and some never do.
“One of the peculiar aspects about this virus is that after you’ve been infected, it takes about five days for you to start to see symptoms [in people that develop symptoms],” remarked Dr. Noggle “There’s usually about a two-day period where you have the virus and are contagious but don’t have symptoms.”
Again, this is why masks are so important. Even if you don’t feel sick, you could very easily be infected and inadvertently transmitting the virus to others, and masks significantly reduce your chances of doing so.

“So, the different tests that are being proposed are aiming to detect the virus as early as possible by looking for the viral genome. But that’s only possible once the virus has replicated enough for you to have enough in your respiratory system that it can be successfully harvested using a test.”
Improved testing technology will be critical for containing the virus, and NYSCF — Robertson Stem Cell Investigator Alumnus Feng Zhang, PhD, is making rapid progress towards a point-of-care COVID-19 test.
If you get the virus and recover, are you immune?
When your body is infected by a virus, your immune system produces antibodies — proteins that counteract the infection and prevent you from being reinfected in the future. Blood tests to detect SARS-CoV-2 antibodies are therefore being widely used to determine who has already had the virus and who might be immune.

“It appears, based on data that’s come out over the past few weeks, that if you successfully mount an antibody response to the virus, those antibodies seem to be protective and will prevent you from being reinfected,” noted Dr. Noggle. “We know from other coronaviruses that immunity can last from one to seventeen years, but it’s still unclear how long protection to SARS-CoV-2 will last.”
More antibody research is needed to understand COVID-19 immunity, and may also give us clues on how to treat or prevent viral infection, which is an active area of study for NYSCF – Robertson Stem Cell Investigator Alumnus Deepta Bhattacharya, PhD.
What causes severe cases of COVID-19?
Our immune systems are designed to sense viral invaders and respond by attacking them, including with antibodies. But in some COVID-19 cases, the immune system kicks into overdrive – a phenomenon called a ‘cytokine storm’ – leading to severe and sometimes fatal consequences.
“It appears that SARS-CoV-2 can trigger an unbalanced immune response in some people,” said Dr. Noggle. “Your immune system will release lots of different molecules called cytokines that have downstream consequences all over the body. It’s basically your immune system attacking almost all different cells or tissues in your body.”
Scientists are working hard to understand what makes certain people vulnerable to the cytokine storm. Several underlying conditions, including heart disease, diabetes, and lung disease, are frequently associated with severe cases, and it is likely that individual genetics play a role as well. By making stem cell models from severely affected patients, we can begin to understand why individual responses to the virus vary.

“There has been previous research to understand this cytokine storm, as well as drugs that try to block it. And these are currently in clinical trials in patients experiencing severe responses,” noted Dr. Noggle. “And with stem cells, we can look at hundreds to thousands of people using their stem cells as biological avatars in the laboratory, allowing us to study how genetics may lead to severe responses and what could be done to treat or prevent them.”
What drugs are being used or developed for COVID-19?
The hunt is on for new or existing drugs that can interfere with the SARS-CoV-2 life cycle or help fight it off. Some try to block the virus’s spike proteins so that it can’t get into cells. Others, like remdesivir, block replication of the viral genome. Another class of drugs try to block the virus from releasing its contents into cells – the most famous being hydroxychloroquine, although this drug can cause critical complications.
“Hydroxychloroquine has been around for a while to treat malaria and different autoimmune diseases, and it’s usually used in a lower dose to accomplish that,” explained Dr. Noggle. “At very high doses, in cell culture models in the lab, it can also block the release of the viral genome into the cell. But it takes really high doses to do that. And one of the problems with hydroxychloroquine is that at high doses, it can interact with receptors in the heart and cause heart problems.”
Convalescent serum — that is, blood samples processed from people who have recovered from a previous COVID-19 infection — is also being tested as a treatment.
“Convalescent serum contains antibodies that have been isolated from people that have already been infected, have recovered, and have mounted a good immune response. They have antibodies in their system that can be injected into people that are currently undergoing disease.”
These are among several therapies being investigated in clinical trials, and it is too soon to be sure which will be effective. Many more drugs are also being discovered and tested using laboratory models, and patient-specific stem cell models are beginning to play a critical role in this process.
How is NYSCF using stem cells to accelerate treatments for COVID-19?
NYSCF is using stem cells to create the actual human cells susceptible to SARS-CoV-2 infection, study how the virus affects them, and find new drugs likely to be effective in humans.
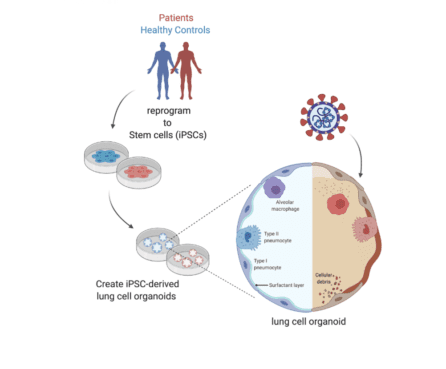
“We are creating the particular types of lung cells that are highly infectible, such as the cells that secrete the mucus that coats your lungs to keep them functioning and healthy,” says Dr. Noggle. “When these cells get infected, they make more of the virus, but they also die – eliminating the source of that mucus and that ability to clear things out of your lungs. So, what we can do is use those cells, created from stem cells, to test large amounts of existing drugs or find new drugs that would prevent the infection in the first place, sparing these cells and stopping the virus from replicating.”
Dr. Noggle’s team at NYSCF is creating these vulnerable cell types as a resource for the COVID-19 research community, and through several active collaborations will test promising drugs identified by other scientists. For example, NYSCF – Robertson Investigator Alumna Shuibing Chen, PhD, of Weill Cornell Medicine is using stem cells to model SARS-CoV-2 infection of ten different types of tissue believed to be most susceptible to infection. She is using these stem cell models to test which drugs can block entry of the virus into cells, and NYSCF is working closely with her to scale up and accelerate her drug screens.

Given NYSCF’s active research efforts in many other disease areas, we are well positioned to study how COVID-19 affects patients with underlying conditions, as well as its long-term consequences on different organs like the heart and brain.
“We’re interested in the long-term consequences of infection in those that have recovered. We’re also trying to understand how patients with the different chronic diseases that we currently study might be impacted. Are there going to be neurological impacts? We know the virus can infect the cells impacted by type 1 diabetes, so how will those patients be affected in the long term?”
Altogether, Dr. Noggle is optimistic that science will bring an end to this pandemic.
“The enthusiasm that my colleagues and researchers around the world have shown in coming together to address this disease has been very encouraging for me,” he remarked. “I look forward to working with all of them to try to find ways to prevent and treat this virus and to return people back to health.”
Learn more about the numerous efforts in the NYSCF community to accelerate solutions for COVID-19, and more on what you need to know about COVID-19 can be found here.
Watch Dr. Noggle’s full presentation below.

