A Cell Therapy in Development at NYSCF Could Give AMD Patients Their Sight Back
For patients with age-related macular degeneration (AMD), a cell therapy in development at the NYSCF Research Institute could provide...For patients with age-related macular degeneration (AMD), a cell therapy in development at the NYSCF Research Institute could provide what sounds like a miracle: the restoration of their sight.

Approximately 11 million people in the US suffer from AMD, a number that is expected to double by 2050, and those over the age of 75 have a nearly 30% risk of contracting the disease. The global cost of treating AMD is $343 billion.
“We are excited to realize the promise of stem cells in our first cell therapy and hopefully offer relief to patients who have lost their sight to AMD,” said NYSCF CEO Susan L. Solomon, JD, in a press release. “This will also be critical groundwork for future cell therapies targeted at other major diseases of our time.”
For the therapy, NYSCF scientists will create healthy retinal pigmented epithelial (RPE) cells (the cells lost in the disease) from a patient’s stem cells, which will be placed onto a specially designed patch and surgically implanted behind the retina by surgeons at Columbia University – NY Presbyterian Hospital led by Stanley Chang, MD, Stephen Tsang, MD, and Tongalp Tezel, MD.
“I am hopeful that this therapy will lead to improving or stabilizing vision for patients with the most common form of macular degeneration,” remarked Dr. Chang. “NYSCF’s approach to creating a personalized therapy is innovative and promising, and I am happy to be a part of this exciting project.”
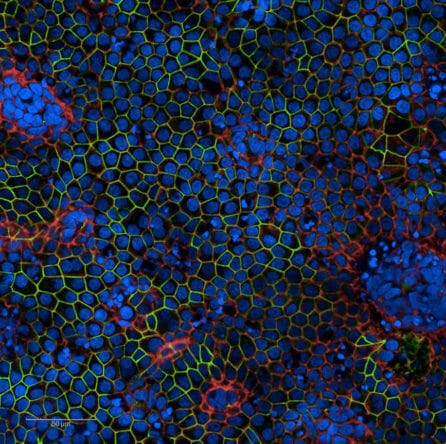
The RPE patches will be created using a method established by Kapil Bharti, PhD, (the National Eye Institute at the National Institutes of Health) that is being used in a clinical trial currently enrolling patients with AMD. CellRay LLC, a fully-owned subsidiary of New York Stem Cell Foundation, Inc., has obtained a license to use Dr. Bharti’s protocol and has executed a collaborative research and development agreement allowing our scientists to leverage Dr. Bharti’s extensive preclinical work to accelerate the clinical trial timeline. This is an ‘autologous’ cell therapy, meaning that it uses a patient’s own cells, decreasing risk of the immune system rejecting the transplant.
“My lab at the NEI has received FDA approval for an autologous iPSC-derived RPE cell therapy for AMD patients. The Phase I/IIa clinical trial is currently ongoing at NIH. We are excited to collaborate with scientists at NYSCF to further this technology,” explained Dr. Bharti. “Using RPE derived from patients’ own blood cells in this therapy will be uniquely advantageous because it should reduce the likelihood of immune rejection, a common issue for transplantations.”
The clinical-grade cells are being produced in NYSCF’s state-of-the-art Good Manufacturing Practices (GMP) facility, which was completed last summer.
“We are looking forward to advancing this therapy toward the clinic,” agreed Howard Kim, PhD, NYSCF Senior Director of Cell Therapy Programs. “With construction of the GMP facility complete and these licenses in place, we are closer than ever to reaching patients in desperate need of new solutions.”
“Macular degeneration is just the beginning – the new facility gives us a dedicated, unique, and safe environment in which to create cell therapies for a variety of diseases, and we look forward to bringing these therapies to patients,” noted Cecile Terrenoire, PhD, NYSCF Senior Director of Process Development.
Read more in a press release
Cover image: Stem-cell-derived RPE cells. Image credit: Cecile Terrenoire, PhD

